Global Rare Neurological Disease Treatment Market Dominated by North America as Germany Emerges with Bayer AG & Pfizer
Germany strengthens its role as a biotech hub, driven by gene therapy, RNA treatments, and precision medicine, shaping neurological care through 2035.
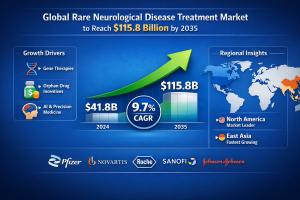
ROCKVILLE, MD, UNITED STATES, April 16, 2026 /EINPresswire.com/ -- According to the latest analysis by Fact.MR, the global rare neurological disease treatment market is projected to grow at a CAGR of 9.8% from 2025 to 2035, driven by rapid advancements in gene therapy, RNA-based therapeutics, and biologics.Get detailed market forecasts, competitive benchmarking, and pricing trends: https://www.factmr.com/connectus/sample?flag=S&rep_id=4594
The market is witnessing multi-billion-dollar incremental opportunity expansion, supported by increasing approvals of high-impact therapies and rising investment in precision medicine.
Germany is emerging as a key European innovation center, benefiting from strong regulatory alignment with the EU, advanced healthcare infrastructure, and growing investments in biotech R&D. The market is transitioning from symptomatic treatments toward disease-modifying and potentially curative therapies, fundamentally reshaping treatment paradigms.
Quick Stats Section
Market Growth (2025–2035): CAGR of 9.8%
Incremental Opportunity: Multi-billion-dollar expansion over forecast period
Leading Segment: Biologics (52.1% share in 2025)
Leading Region: North America; Germany among top EU innovation hubs
Key Players: Pfizer Inc., Novartis AG, F. Hoffmann-La Roche Ltd., Bayer AG, Sanofi S.A., AbbVie Inc., Merck & Co., Johnson & Johnson
Executive Insight for Decision Makers
Germany’s rare neurological treatment landscape is undergoing a paradigm shift toward precision and curative therapies:
Strategic Shift:
From chronic symptom management to gene editing, RNA therapies, and personalized medicine
What Stakeholders Must Do:
Invest in gene therapy pipelines and biomarker-driven diagnostics
Strengthen clinical trial capabilities and AI-driven drug discovery
Collaborate with regulators for accelerated approvals and reimbursement models
Risks of Not Adapting:
Companies failing to transition toward advanced biologics and personalized therapies risk losing relevance as traditional small-molecule treatments decline in effectiveness and demand.
Market Dynamics
Key Growth Drivers
Rising investment in gene and RNA-based therapies
Increasing prevalence of neurodegenerative and rare genetic disorders
Strong regulatory incentives under orphan drug frameworks
Advancements in CRISPR and precision medicine technologies
Key Restraints
Extremely high treatment costs (USD 1–2 million per dose)
Limited reimbursement coverage and accessibility challenges
Complex manufacturing and supply chain requirements
Emerging Trends
Rapid expansion of CRISPR-based gene editing therapies
Integration of AI in drug discovery and clinical trials
Growth of biomarker-driven personalized medicine
Adoption of outcome-based pricing and reimbursement models
Segment Analysis
Leading Segment:
Biologics hold 52.1% market share (2025) due to targeted efficacy and innovation
Fastest-Growing Segment:
Gene therapy and RNA-based therapeutics
By Disease Type:
Neurodegenerative disorders (ALS, Huntington’s, Parkinson’s) dominate with strong R&D focus
Strategic Importance:
Advanced therapies are shifting the market toward potential cures rather than long-term disease management, significantly increasing value per patient.
Supply Chain Analysis (Critical Insight)
Germany plays a central role in Europe’s biopharmaceutical supply chain:
Supply Chain Structure
Raw Material Suppliers:
Biochemical inputs, viral vectors, nucleotides, biologic intermediates
Manufacturers / Producers:
Biopharmaceutical companies producing gene therapies, monoclonal antibodies, and RNA drugs
Research & Development:
Biotech firms, academic institutions, clinical research organizations
Distributors:
Specialty pharma distributors, hospital networks
End-Users:
Hospitals and specialty clinics
Neurology treatment centers
Patients with rare neurological conditions
“Who Supplies Whom”
Biotech suppliers → Pharma manufacturers → Clinical providers → Patients
Research institutions → Support pharma companies → Accelerate drug development
Germany’s ecosystem is defined by strong integration between research, manufacturing, and clinical delivery systems.
Pricing Trends
Pricing Model:
Premium, high-cost therapies with value-based pricing structures
Key Influencing Factors:
R&D and clinical trial costs
Regulatory approvals and exclusivity
Manufacturing complexity
Reimbursement frameworks
Margin Insights:
High margins for innovators due to patent protection and orphan drug incentives
Increasing pressure for outcome-based pricing models
Regional Analysis
Top Growth Markets
United States – 10.7% CAGR
China – Fastest emerging growth
Europe (including Germany) – Strong innovation-driven growth
Germany Focus
Leading European hub for biotech innovation and clinical research
Strong adoption of advanced therapies under EMA frameworks
Increasing investment in AI-driven drug discovery and precision medicine
Developed vs Emerging Markets
Developed Markets (Germany, U.S.):
High innovation, strong regulatory frameworks, premium pricing
Emerging Markets (China, Asia-Pacific):
Rapid expansion, increasing access, growing domestic biotech capabilities
Competitive Landscape
Market Structure: Moderately consolidated with strong presence of global pharma leaders
Key Players
Pfizer Inc.
Novartis AG
F. Hoffmann-La Roche Ltd.
Bayer AG
Sanofi S.A.
AbbVie Inc.
Merck & Co.
Johnson & Johnson Services, Inc.
Teva Pharmaceutical Industries Ltd.
Competitive Strategies
R&D Investment: Focus on gene and RNA therapies
Strategic Partnerships: Collaborations with biotech firms and research institutions
Regulatory Strategy: Leveraging orphan drug incentives
Pipeline Expansion: Focus on neurodegenerative diseases
Strategic Takeaways
For Manufacturers
Accelerate development of gene and cell therapies
Invest in scalable biologics manufacturing platforms
For Investors
Focus on companies with strong R&D pipelines and regulatory approvals
Target precision medicine and AI-driven drug discovery platforms
For Marketers / Distributors
Build expertise in specialty drug commercialization
Strengthen relationships with healthcare providers and payers
Future Outlook
Germany’s rare neurological disease treatment market is expected to:
Lead Europe in precision medicine and advanced therapy adoption
Witness widespread use of gene editing and regenerative medicine by 2035
Benefit from integration of AI and digital health technologies
The long-term outlook indicates a shift toward curative treatments, significantly transforming patient outcomes and healthcare economics.
Conclusion
Germany is emerging as a critical innovation hub in the global rare neurological disease treatment market, supported by cutting-edge research, regulatory support, and strong healthcare infrastructure.
For decision-makers, the opportunity lies in embracing next-generation therapies, forming strategic collaborations, and aligning with evolving reimbursement models to capture long-term value.
Why This Market Matters
The rare neurological disease treatment market represents a transformational shift in modern medicine, moving from symptom management to precision-driven, potentially curative therapies.
Germany’s leadership underscores the importance of innovation, collaboration, and regulatory alignment in unlocking life-changing treatments and shaping the future of global healthcare.
Unlock 360° insights for strategic decision making and investment planning: https://www.factmr.com/checkout/4594
To View Our Related Report:
Rare Hemophilia Factors Market: https://www.factmr.com/report/rare-hemophilia-factors-market
Rare Musculoskeletal Disorder Treatments Market: https://www.factmr.com/report/rare-musculoskeletal-disorder-treatments-market
Rare Disease Genetic Testing Market: https://www.factmr.com/report/rare-disease-genetic-testing-market
Rare Disease Small-Batch CDMO Market: https://www.factmr.com/report/rare-disease-small-batch-cdmo-market
S. N. Jha
Fact.MR
+1 628-251-1583
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.